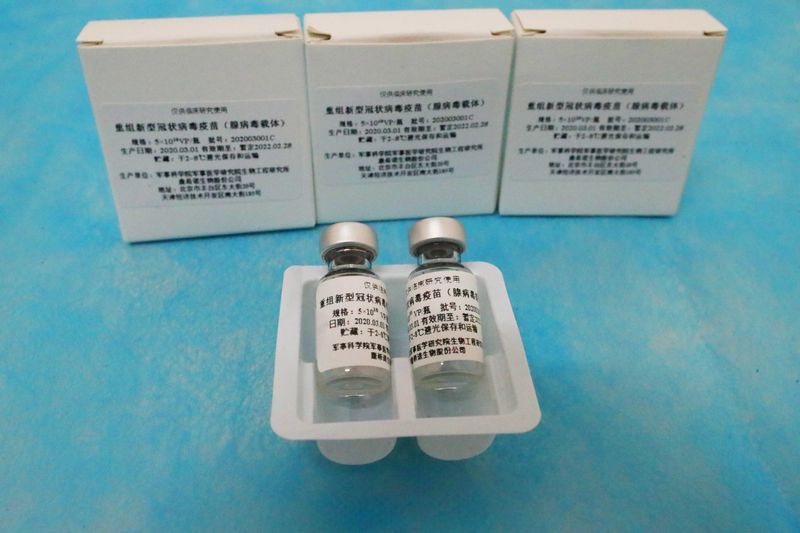
MOSCOW/BEIJING (Reuters) – Russia’s Petrovax has started a late-stage trial of a COVID-19 candidate vaccine from CanSino Biologics Inc in the country, records show, as the Chinese drug firm steps up testing abroad to close in on regulatory approval.
The Ad5-nCoV vaccine already has approval for use by China’s military after early and mid-stage trials, and further late-stage trials are being lined up for Mexico and Saudi Arabia.
CanSino last month said it was in talks for opportunities to launch late-stage – or Phase 3 – trials in Saudi Arabia, Russia, Brazil and Chile.
The company has won a patent approval from Beijing for the vaccine, Chinese state media reported on Sunday, citing documents from the country’s intellectual property regulator.
This is the first trial of a potential vaccine developed by a foreign company to take place in Russia, while the country prepares to start mass production of its domestic shot developed by Moscow’s Gamaleya Institute.
Russian pharmaceutical company Petrovax confirmed it was running the trial for CanSino in a statement on Saturday.
If the vaccine is approved in the country, it said it would also produce the shot at its facilities in the Moscow region, with plans to supply the Russian market, as well as CIS countries. It didn’t give further details.
CanSino and compatriots including Sinovac Biotech Ltd and China National Pharmaceutical Group Co Ltd (Sinopharm) are testing candidates overseas as China becomes less conducive for Phase 3 trials due to its small number of new COVID-19 cases.
The completion of Phase 3 trials is generally required for regulatory approval to distribute a vaccine for public use.
Russia’s state register https://bit.ly/2DSHY0O for clinical trials showed a Phase 3 study of CanSino’s potential shot, expected to recruit 625 participants across eight medical institutes to test the safety and effectiveness of Ad5-nCoV, began on Friday.
CanSino declined to offer more details on the Russian trial when contacted by Reuters, saying the information was confidential.
Last month, Russian biotech group Biocad said it was discussing producing in China a potential COVID-19 vaccine being developed by Russia’s Vector state virology institute.
With 927,745 cases, Russia has the world’s fourth-highest number of infections.
(Reporting by Polina Ivanova and Gabrielle Tetrault-Farber in Moscow and Roxanne Liu in Beijing; Editing by Josephine Mason and Mark Potter)